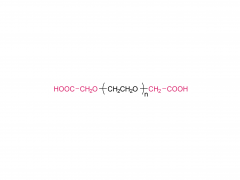
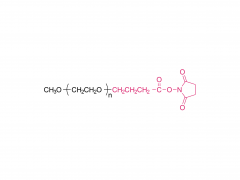
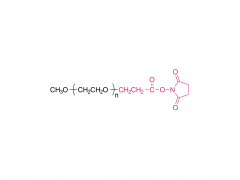
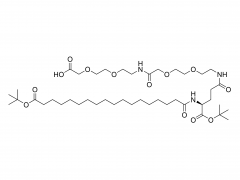
Process Biochemistry. Volume 52, January 2017, Pages 183-191 https://doi.org/10.1016/j.procbio.2016.09.029 Effect of protein immunogenicity and PEG size and branching on the anti-PEG immune response to PEGylated proteins Xue Wan, Juankun Zhang, Weili Yu, Lijuan Shen, Shaoyang Ji, Tao Hu Abstract PEGylation has successfully improved the pharmacological properties of therapeutic proteins. However, polyethylene glycol (PEG) has been burdened by immunogenicity that renders a negative clinical effect on therapeutic proteins. The anti-PEG immune response to PEGylated proteins possibly depends on the nature of proteins and the conjugated methoxy PEG (mPEG). Thus, it is necessary to investigate the effects of protein immunogenicity, the extent of PEGylation, the molecular weight (Mw), and the branching of mPEG on the anti-PEG immune response. Ovalbumin, tetanus toxoid (TT), TT–TT conjugate, and TT–bovine serum albumin conjugate were used as target proteins. PEGylated proteins with different extents of PEGylation were obtained by fractionation of the PEGylated TT with size exclusion chromatography. The PEGylated proteins with different Mw and branching of mPEG were obtained by modification of TT with linear mPEG (5 kDa and 20 kDa) and branched mPEG (20 kDa). The PEGylated proteins elicited high levels of anti-PEG antibodies (predominantly IgM and IgG1). The anti-PEG immune response depended on the immunogenicity of proteins, the extent of PEGylation, and the Mw of mPEG. In contrast, branching of mPEG had an insignificant effect on the anti-PEG immune response to the PEGylated proteins. Related products Abbreviation: mPEG-MAL Name: Methoxypoly(ethylene glycol) maleimide For more product information, please contact us at: US Tel: 1-844-782-5734 US Tel: 1-844-QUAL-PEG CHN Tel: 400-918-9898 Email: sales@sinopeg.com
View More