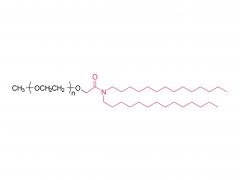
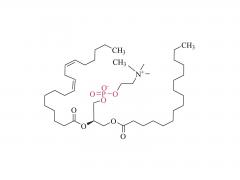
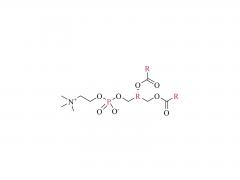
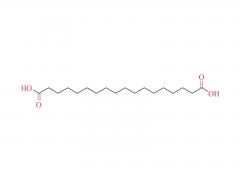
I. Product Overview
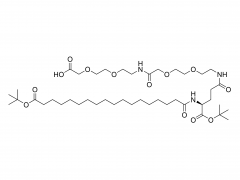
Fmoc-PEGn-COOH is a multifunctional heterobifunctional crosslinker that integrates a protecting group, a hydrophilic spacer arm, and a reactive terminal. The molecule features an Fmoc protecting group at one end, a reactive carboxylic acid at the other, and a polyethylene glycol (PEG) chain of variable length in the middle. This unique design makes it an indispensable tool in chemical biology, drug delivery, and novel drug development (e.g., PROTACs).
II. Structural Features
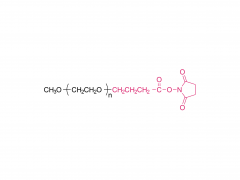
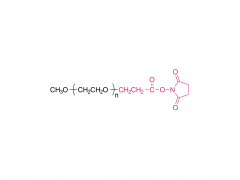
Fmoc-protected amino terminus (N-terminus): Provides stable amino protection to prevent undesirable side reactions. Can be rapidly and efficiently removed under mild basic conditions to expose a free amino group (-NH₂), facilitating subsequent coupling with carboxyl groups or activated esters.
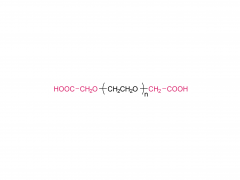
Tunable PEG chain (middle section): The flexible length of the PEG chain (n=1-12) allows precise modulation of molecular distance and linker length. It imparts excellent hydrophilicity and water solubility, effectively improving the biocompatibility of conjugated molecules and reducing aggregation. It also provides conformational flexibility, helping the linked moieties maintain their active conformations, and enhances overall stability against enzymatic degradation.
Reactive carboxylic acid terminus (C-terminus): Can be activated (e.g., with EDC, HATU, DIC) to react with amino groups (-NH₂) to form stable amide bonds, enabling conjugation to peptides, proteins, small molecules, or solid supports. This is the core site for further functionalization.
III. Key Application Areas
Solid-Phase Peptide Synthesis (SPPS): Serves as a cleavable linker to anchor peptide chains to resin. After synthesis, Fmoc deprotection and cleavage yield C-terminally modified peptides. Used to introduce PEG spacers into peptide chains, improving peptide solubility and biophysical properties.
Bioconjugation & Drug Delivery: Links drug molecules to targeting ligands (e.g., antibodies, peptides, small molecules) to construct antibody-drug conjugates (ADCs) or targeted delivery systems. Used to modify proteins, nanoparticles, or material surfaces, introducing Fmoc-protected active sites for subsequent click chemistry or further conjugation.
Materials Science & Surface Chemistry: Functionalizes gold nanoparticles, quantum dots, silica microspheres, etc., to build biosensing interfaces. Introduces PEG brushes onto material surfaces to create bioinert surfaces that resist non-specific protein adsorption.
IV. Key Applications in PROTAC Technology
PROTAC (Proteolysis-Targeting Chimera) is a revolutionary targeted protein degradation technology. Fmoc-PEGn-COOH plays a critical role as a "linker precursor" or "cleavable linker" in this context.
As a key building block for PROTAC synthesis:
Modular synthesis: Fmoc-PEGn-COOH serves as the bridge connecting the E3 ubiquitin ligase ligand and the target protein ligand. Its carboxylic acid end is typically coupled to an amino group on the E3 ligand; after Fmoc removal, the exposed amino group is coupled to a carboxyl group on the target protein ligand, enabling efficient assembly of the complete PROTAC molecule.
Structure-activity relationship studies: By using PEG chains of different lengths (n=1-12), researchers can systematically investigate the impact of linker length on ternary complex formation efficiency, degradation activity, and cell permeability. An optimal PEG length helps maintain the ideal spatial orientation between the two ligands.
Improving the drug-like properties of PROTAC molecules:
Enhances water solubility: Many PROTAC molecules suffer from poor solubility due to high hydrophobicity. Introducing a hydrophilic PEG chain significantly improves their water solubility and dispersibility in cell culture media, facilitating in vitro activity assays.
Modulates membrane permeability: The length and properties of the PEG chain influence the cell permeability of PROTACs; short PEG chains help balance hydrophilicity and membrane permeability.