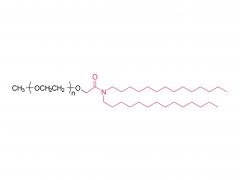
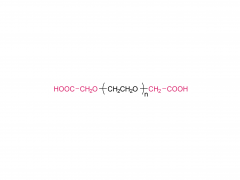
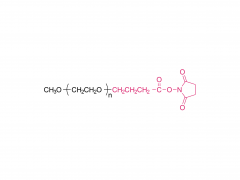
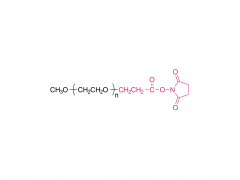
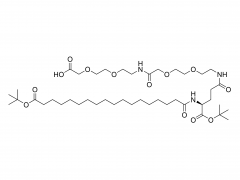
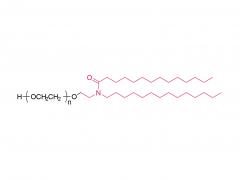
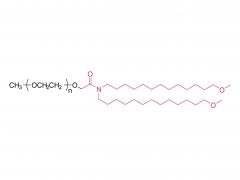
Heptadecan-9-yl 8-((2-hydroxyethyl) (6-oxo-6-(undecyloxy) hexyl) amino) octanoate)
Has been registered, DMF 038143, CDE F20210000505
Custom synthesis & CMO services are available.
License is required from license holder.
SINOPEG is serving pharmaceutical and medical device companies around the globe, with product presence in various pharmaceutical/device development pipeline (pre-clinical, clinical, and post authorization large scale supply). Our facility is ISO9001 and ISO13485 certified, and is operating according to ICH Q7A guidelines to produce products for pharmaceutical companies.
Please contact us at sales@sinopeg.com for PEG derivatives. Our online catalog or inventory may not listed or have all molecular weights and functional groups, which may be available by custom synthesis. Please contact us at sales@sinopeg.com for quotation and availability.
Reference:
1. Wan J, Wang C, Wang Z, et al. CXCL13 promotes broad immune responses induced by circular RNA vaccines. Proc Natl Acad Sci U S A. 2024;121(44):e2406434121. doi:10.1073/pnas.2406434121
2. Wang L, Wan J, He W, et al. IL-7 promotes mRNA vaccine-induced long-term immunity. J Nanobiotechnology. 2024;22(1):716. Published 2024 Nov 16. doi:10.1186/s12951-024-02993-5
3. Tong H, Ma Z, Yu J, et al. Optimizing Peptide-Conjugated Lipid Nanoparticles for Efficient siRNA Delivery across the Blood-Brain Barrier and Treatment of Glioblastoma Multiforme. ACS Chem Biol. 2025;20(4):942-952. doi:10.1021/acschembio.5c00039
4. Shao X, Liu L, Weng C, et al. Leveraging an mRNA Platform for the Development of Vaccines Against Egg Allergy. Vaccines (l). 2025;13(5):448. Published 2025 Apr 24. doi:10.3390/vaccines13050448
5. Deng X, Yang Y, Gan L, et al. Engineering Lipid Nanoparticles to Enhance Intracellular Delivery of Transforming Growth Factor-Beta siRNA (siTGF-β1) via Inhalation for Improving Pulmonary Fibrosis Post-Bleomycin Challenge. Pharmaceutics. 2025;17(2):157. Published 2025 Jan 24. doi:10.3390/pharmaceutics17020157
6. Zhu C, Roa N, Neathery E et al. Comparative technical and operational assessment of current and emerging bench-scale lipid nanoparticle platforms for production of mRNA vaccines [version 1]. VeriXiv 2025, 2:96 (https://doi.org/10.12688/verixiv.982.1)
7. Ye T, Zhou J, Guo C, et al. Polyvalent mpox mRNA vaccines elicit robust immune responses and confer potent protection against vaccinia virus. Cell Rep. 2024;43(6):114269. doi:10.1016/j.celrep.2024.114269
8. Cheng X, Zheng X, Tao K, et al. Freezing induced incorporation of betaine in lipid nanoparticles enhances mRNA delivery. Nat Commun. 2025;16(1):4700. Published 2025 May 20. doi:10.1038/s41467-025-60040-9
9. Liu S, Wen Y, Shan X, et al. Charge-assisted stabilization of lipid nanoparticles enables inhaled mRNA delivery for mucosal vaccination. Nat Commun. 2024;15(1):9471. Published 2024 Nov 2. doi:10.1038/s41467-024-53914-x
10. Kozlova A, Pateev I, Shepelkova G, et al. A Cap-Optimized mRNA Encoding Multiepitope Antigen ESAT6 Induces Robust Cellular and Humoral Immune Responses Against Mycobacterium tuberculosis. Vaccines (Basel). 2024;12(11):1267. Published 2024 Nov 9. doi:10.3390/vaccines12111267
11. Wang H, Peng Q, Dai X, et al. A SARS-CoV-2 EG.5 mRNA vaccine induces a broad-spectrum immune response in mice. MedComm (2020). 2025;6(1):e779. Published 2025 Jan 2. doi:10.1002/mco2.779
12. Cao H, Zhang X, Cheng J, et al. A QS21 + CpG-Adjuvanted Trivalent HSV-2 Vaccine and Trivalent HSV-2 mRNA Vaccine Induce a Strong Immune Response, Protect Against HSV-2 Infection, and Cross-Protect Against HSV-1 Infection in Mice. Vaccines (Basel). 2025;13(5):497. Published 2025 May 6. doi:10.3390/vaccines13050497
13. Zhang S, Wang X, Zhao T, Yang C, Huang L. Development and Evaluation of the Immunogenic Potential of an Unmodified Nucleoside mRNA Vaccine for Herpes Zoster. Vaccines (Basel). 2025;13(1):68. Published 2025 Jan 13. doi:10.3390/vaccines13010068
14. Wang Z, Tian C, Zhu J, et al. Avian influenza mRNA vaccine encoding hemagglutinin provides complete protection against divergent H5N1 viruses in specific-pathogen-free chickens. J Nanobiotechnology. 2025;23(1):55. Published 2025 Jan 29. doi:10.1186/s12951-025-03156-w
15. Widayat, Wahyu & Hardianto, Ari & Hidayati, Rahma & Neni, Nurainy & Burhanudin, Muhammad & Yusuf, Muhammad & Subroto, Toto. (2025). The Effect of Mixed Lipid Concentrations and Sucrose on the Size of the Lipid Nanoparticles Containing mRNAs. Trends in Sciences. 22. 8985. 10.48048/tis.2025.8985.
16. Jeon JH, Kim S, Kim SY, et al. Heterologous prime-boost vaccination drives stromal activation and adaptive immunity against SARS-CoV-2 variants. Front Immunol. 2025;16:1597417. Published 2025 May 28. doi:10.3389/fimmu.2025.1597417
17. Tong H, Ma Z, Li D, et al. Tuning the polyethylene glycol-lipid anchor length of lipid nanoparticles to enhance brain-targeted siRNA delivery. Acta Biomater. Published online October 11, 2025. doi:10.1016/j.actbio.2025.10.018
18. Xiao C, Liu X, Liu P, et al. Oncogenic roles of young human de novo genes and their potential as neoantigens in cancer immunotherapy. Cell Genom. 2025;5(9):100928. doi:10.1016/j.xgen.2025.100928
19. Zheng X, Liu B, Ni P, et al. Development and application of an uncapped mRNA platform. Ann Med. 2025;57(1):2437046. doi:10.1080/07853890.2024.2437046
20. Liu D, Wang X, Xu L, et al. Screening lipid nanoparticles using DNA barcoding and qPCR. Colloids Surf B Biointerfaces. 2025;251:114598. doi:10.1016/j.colsurfb.2025.114598
21. Abidin, Muhamad & Yusuf, Muhammad & Widayat, Wahyu & Subroto, Toto & Neni, Nurainy & Hardianto, Ari. (2023). The Implementation of Response Surface Methodology in the Optimization of Lipid Nanoparticle Preparation for Vaccine Development. Trends in Sciences. 21. 7142. 10.48048/tis.2024.7142.
22. Li W, Li Y, Li J, et al. All-Trans-Retinoic Acid-Adjuvanted mRNA Vaccine Induces Mucosal Anti-Tumor Immune Responses for Treating Colorectal Cancer. Adv Sci (Weinh). 2024;11(22):e2309770. doi:10.1002/advs.202309770
23. Li M, Yi J, Lu Y, et al. Modified PEG-Lipids Enhance the Nasal Mucosal Immune Capacity of Lipid Nanoparticle mRNA Vaccines. Pharmaceutics. 2024;16(11):1423. Published 2024 Nov 7. doi:10.3390/pharmaceutics16111423
24. Yu W, Chen X, Chen Q, et al. A Safe and Broad-spectrum SARS-CoV-2 mRNA Vaccine with a New Delivery System for In-situ Expression. Virol Sin. Published online September 4, 2025. doi:10.1016/j.virs.2025.09.001
25. Li H, Hu Y, Li J, et al. Intranasal prime-boost RNA vaccination elicits potent T cell response for lung cancer therapy. Signal Transduct Target Ther. 2025;10(1):101. Published 2025 Mar 24. doi:10.1038/s41392-025-02191-1
26. Yi J, Lu Y, Liu N, et al. Chitosan and mannose-modified dual-functional mRNA-LNP vaccines for robust systemic and mucosal immune responses. J Control Release. 2025;384:113891. doi:10.1016/j.jconrel.2025.113891
27. Shi R, Liu X, Wang Y, et al. Long-term stability and immunogenicity of lipid nanoparticle COVID-19 mRNA vaccine is affected by particle size. Hum Vaccin Immunother. 2024;20(1):2342592. doi:10.1080/21645515.2024.2342592
28. Lu Y, Yang Y, Yi J, et al. Design, optimization, and evaluation of lyophilized lipid nanoparticles for mRNA-based pulmonary mucosal vaccination. Mater Today Bio. 2025;32:101813. Published 2025 May 4. doi:10.1016/j.mtbio.2025.101813
29. Lv K, Yu Z, Wang J, et al. Discovery of Ketal-Ester Ionizable Lipid Nanoparticle with Reduced Hepatotoxicity, Enhanced Spleen Tropism for mRNA Vaccine Delivery. Adv Sci (Weinh). 2024;11(45):e2404684. doi:10.1002/advs.202404684
30. Lee Y, Jeong M, Lee G, et al. Development of Lipid Nanoparticle Formulation for the Repeated Administration of mRNA Therapeutics. Biomater Res. 2024;28:0017. Published 2024 May 22. doi:10.34133/bmr.0017
31. Na GS, Joo JU, Lee JY, et al. Full-cycle study on developing a novel structured micromixer and evaluating the nanoparticle products as mRNA delivery carriers. J Control Release. 2024;373:161-171. doi:10.1016/j.jconrel.2024.07.019
32. Dong L, Wang J, Xia Q. Chemically modified tRNA enhances the translation capacity of mRNA rich in cognate codons. Nat Commun. 2025;16(1):7825. Published 2025 Aug 22. doi:10.1038/s41467-025-62981-7
33. Wan J, Wang Z, Wang L, et al. Circular RNA vaccines with long-term lymph node-targeting delivery stability after lyophilization induce potent and persistent immune responses. mBio. 2024;15(1):e0177523. doi:10.1128/mbio.01775-23
34. Tang S, Huang L, Ge J, et al. Influence of salt solution on the physicochemical properties and in vitro/ in vivo expression of mRNA/LNP. J Nanobiotechnology. 2025;23(1):223. Published 2025 Mar 19. doi:10.1186/s12951-025-03318-w
35. Yu, Juntao & Li, Qian & Luo, Shenggen & Wang, Xiaona & Cheng, Qiang & hu, Rongkuan. (2024). Targeted LNPs deliver mRNA encoding IL-15 superagonists to balance efficacy and toxicity in cancer therapy. 10.1101/2024.01.11.575299.
36. Sun, Zhen & Liu, Yuxiao & Zhang, Haoyi & Ge, Ting & Pan, Yuting & Liu, Yang & Wu, Miaomiao & Shan, Tao & Wu, Qi & Zhu, Guoqiang & Chen, Kangming. (2025). Next-Generation saRNA Platforms: Systematic Screening and Engineering Enhances Superior Protein Expression and Organ-Specific Targeting for RNA Therapeutics. 10.1101/2025.03.30.644708.
37. Luo C, Li Y, Liu H, et al. Intracellular trafficking of lipid nanoparticles is hindered by cholesterol. Int J Pharm. 2025;671:125240. doi:10.1016/j.ijpharm.2025.125240
38. Chen, Xiaoxia & Li, Mengrong & Jiang, Fan & Hong, Liang & Liu, Zhuo. (2024). Revealing a Correlation between Structure and in vitro Activity of mRNA Lipid Nanoparticles. 10.1101/2024.09.01.610730.
39. Liu X, Li Z, Li X, et al. A single-dose circular RNA vaccine prevents Zika virus infection without enhancing dengue severity in mice. Nat Commun. 2024;15(1):8932. Published 2024 Oct 16. doi:10.1038/s41467-024-53242-0
40. Joo, Jeong-Un & Na, Gi-Su & Sharma, Vikas & Mottafegh, Amirreza & Kim, Dongpyo. (2024). Ultrafast continuous flow-dialysis for nanoparticle-based drug delivery systems via microfluidic-multiple buffer injector. Chemical Engineering Journal. 500. 156631. 10.1016/j.cej.2024.156631.
41. Ma X, Liu S, Zhang S, et al. Engineering of mRNA vaccine platform with reduced lipids and enhanced efficacy. Nat Commun. 2025;16(1):8913. Published 2025 Oct 7. doi:10.1038/s41467-025-63965-3
42. Chen YC, Lee YL, Lee CA, Lin TY, Hwu EE, Cheng PC. Development of a Lipid-encapsulated TGFβRI-siRNA Drug for Liver Fibrosis Induced by Schistosoma mansoni. PLoS Negl Trop Dis. 2024;18(9):e0012502. Published 2024 Sep 12. doi:10.1371/journal.pntd.0012502
43. Francis JA, Wright L, Oerlemans SK, van Wegen R, Falconer RJ. Idiosyncratic behaviour of pegylated lipid nanoparticles used in RNA delivery: A dynamic light scattering study. Int J Pharm. Published online September 20, 2025. doi:10.1016/j.ijpharm.2025.126196
44. Ren L, Zhao Z, Chao Y, et al. Optimization of Lipid Nanoparticles with Robust Efficiency for the Delivery of Protein Therapeutics to Augment Cancer Immunotherapy. Adv Sci (Weinh). 2025;12(17):e2500844. doi:10.1002/advs.202500844
45. Chen, Xiaoxia & Ye, Yongfeng & Li, Mengrong & Zuo, Taisen & Xie, Zhenhua & Ke, Robin & Cheng, He & Hong, Liang & Liu, Zhuo. (2024). Structural Characterization of mRNA Lipid Nanoparticles in the Presence of Intrinsic Drug-free Lipid Nanoparticles. 10.1101/2024.09.27.614859.
46. Wang L, Wan J, He W, et al. IL-7 promotes mRNA vaccine-induced long-term immunity. J Nanobiotechnology. 2024;22(1):716. Published 2024 Nov 16. doi:10.1186/s12951-024-02993-5