Lipid nanoparticles (LNP) can play an important role in gene editing therapy. IntelliaTherapeutics and Regeneron recently announced that their co-development program, ntra-2001, a systemic CRISPR/Cas9 therapy, has achieved positive results in a phase I clinical trial. A single dose of NTLA2001 resulted in an average 87% decrease in serum transthyroxine protein level (TTR), with a maximum reduction of 96% at day 28.CRISPR/Cas9 is a gene-editing tool that makes permanent, precisely targeted changes to a patient's chromosome and fixes potential genetic mutations.Ntra-2001 is a CRISPR/ CAS9-based treatment for hereditary transthyroxine protein-mediated amyloidosis with polyneuropathy (ATTRV-PN).
According to Intellia Therapeutics, NTRA-2001 is a targeted delivery of LNP in humans that selectively knocks out disease-causing genes and restores necessary genetic function through targeted insertion. Three of the six patients treated in the Phase I trial received a dose of 0.1mg/kg ntLA-2001 and the other three received a dose of 0.3mg/kg NTLA2001.At day 28, TTR decreased by an average of 52% in patients receiving 0.1mg/kg and 87% in patients receiving 0.3mg/kg, with a 97% reduction in one patient.
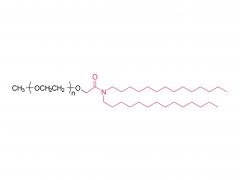
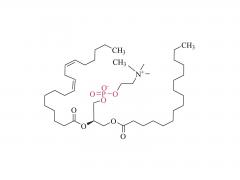
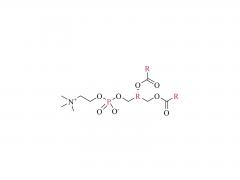
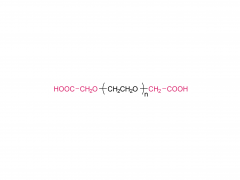
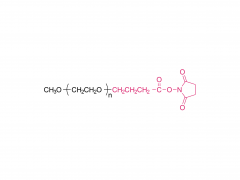
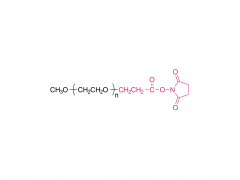
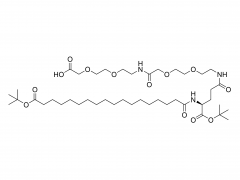
As revealed in IntelliaTherapeutics' patent, LNP contains amine lipids for encapsulation and in vivo escape, neutral and helper lipids for stabilization, and cloaking lipids. In general, LNPS used on CRISPR/Cas9 include DSPC, cholesterol, PE2K-DMG and other liposomes, which are mostly similar to those used for LNPS of mRNA vaccines.
XIAMEN SINOPEG BIOTECH CO., LTD. has been developing DDS sustained-release system for more than ten years, and has strong technical reserves and experienced quality team. The high-quality polyethylene glycol derivatives developed and produced by the company have been successfully applied in the long-acting modification of PEG proteins, peptides and three types of medical devices. We also supply high purity fatty acid side chain to the market for peptide modification. In recent years, SINOPEG has turned to polyethylene glycol phospholipid, polyethylene glycol block copolymer and other high-end complex preparations, and has carried out a number of projects with domestic leading pharmaceutical enterprises. Interested friends can contact us through the following ways:
US Tel: 1-844-782-5734
CHN Tel: 400-918-9898
Email: sales@sinopeg.com
Reference
1. Intellia Therapeutics. (2021).Source: retrieve Intellia Therapeutics: https://www.intelliatx.com/crisprcas9/types-of-edits-2/
2.IntelliaTherapeutics(NTLA.us) and Regentium (RegN.us) announce the results of the first CRISPR clinical trial.Retrieve source: baidu: https://baijiahao.baidu.com/s?id=1703680441595581467&wfr=spider&for=pc