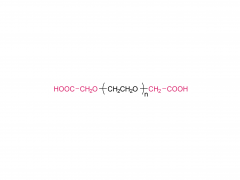
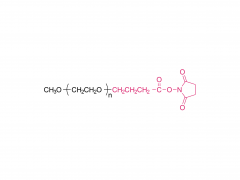
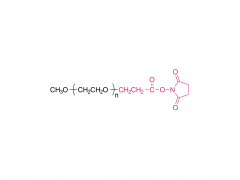
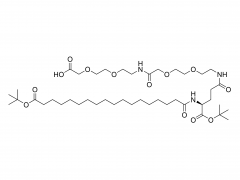
Int J Nanomedicine. 2015 Aug 20:10:5219-35. doi: 10.2147/IJN.S82847. eCollection 2015. D-α-tocopherol polyethylene glycol succinate-based derivative nanoparticles as a novel carrier for paclitaxel delivery Yupei Wu, Qian Chu, Songwei Tan, Xiangting Zhuang, Yuling Bao, Tingting Wu, Zhiping Zhang Abstract Paclitaxel (PTX) is one of the most effective antineoplastic drugs. Its current clinical administration Taxol(®) is formulated in Cremophor EL, which causes serious side effects. Nanoparticles (NP) with lower systemic toxicity and enhanced therapeutic efficiency may be an alternative formulation of the Cremophor EL-based vehicle for PTX delivery. In this study, novel amphipathic 4-arm-PEG-TPGS derivatives, the conjugation of D-α-tocopherol polyethylene glycol succinate (TPGS) and 4-arm-polyethylene glycol (4-arm-PEG) with different molecular weights, have been successfully synthesized and used as carriers for the delivery of PTX. These 4-arm-PEG-TPGS derivatives were able to self-assemble to form uniform NP with PTX encapsulation. Among them, 4-arm-PEG(5K)-TPGS NP exhibited the smallest particle size, highest drug-loading efficiency, negligible hemolysis rate, and high physiologic stability. Therefore, it was chosen for further in vitro and in vivo investigations. Facilitated by the effective uptake of the NP, the PTX-loaded 4-arm-PEG(5K)-TPGS NP showed greater cytotoxicity compared with free PTX against human ovarian cancer (A2780), non-small cell lung cancer (A549), and breast adenocarcinoma cancer (MCF-7) cells, as well as a higher apoptotic rate and a more significant cell cycle arrest effect at the G2/M phase in A2780 cells. More importantly, PTX-loaded 4-arm-PEG(5K)-TPGS NP resulted in a significantly improved tumor growth inhibitory effect in comparison to Taxol(®) in S180 sarcoma-bearing mice models. This study suggested that 4-arm-PEG(5K)-TPGS NP may have the potential as an anticancer drug delivery system. Keywords: 4-arm-PEG; TPGS; antitumor; nanoparticles; paclitaxel. Related products Abbreviation: 4-arm-PEG For more product information, please contact us at: US Tel: 1-844-782-5734 US Tel: 1-844-QUAL-PEG CHN Tel: 400-918-9898 Email: sales@sinopeg.com
View More