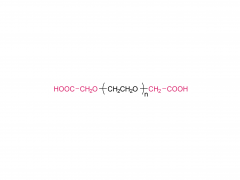
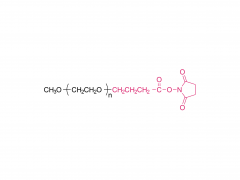
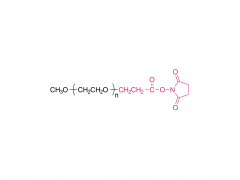
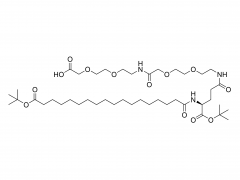
Int J Nanomedicine. 2017 Aug 16;12:5863-5877. doi: 10.2147/IJN.S141982. eCollection 2017. pH-sensitive and folic acid-targeted MPEG-PHIS/FA-PEG-VE mixed micelles for the delivery of PTX-VE and their antitumor activity Yan Di 1, Ting Li 1, Zhihong Zhu 1, Fen Chen 2, Lianqun Jia 2, Wenbing Liu 3, Xiumei Gai 1, Yingying Wang 1, Weisan Pan 1, Xinggang Yang 1 Abstract The aim of this study was to simultaneously introduce pH sensitivity and folic acid (FA) targeting into a micelle system to achieve quick drug release and to enhance its accumulation in tumor cells. Paclitaxel-(+)-α-tocopherol (PTX-VE)-loaded mixed micelles (PHIS/FA/PM) fabricated by poly(ethylene glycol) methyl ether-poly(histidine) (MPEG-PHIS) and folic acid-poly(ethylene glycol)-(+)-α-tocopherol (FA-PEG-VE) were characterized by dynamic light scattering and transmission electron microscopy (TEM). The mixed micelles had a spherical morphology with an average diameter of 137.0±6.70 nm and a zeta potential of -48.7±4.25 mV. The drug encapsulation and loading efficiencies were 91.06%±2.45% and 5.28%±0.30%, respectively. The pH sensitivity was confirmed by changes in particle size, critical micelle concentration, and transmittance as a function of pH. MTT assay showed that PHIS/FA/PM had higher cytotoxicity at pH 6.0 than at pH 7.4, and lower cytotoxicity in the presence of free FA. Confocal laser scanning microscope images demonstrated a time-dependent and FA-inhibited cellular uptake. In vivo imaging confirmed that the mixed micelles targeted accumulation at tumor sites and the tumor inhibition rate was 85.97%. The results proved that the mixed micelle system fabricated by MPEG-PHIS and FA-PEG-VE is a promising approach to improve antitumor efficacy. Keywords: drug delivery; folic acid targeting; in vivo antitumor activity; mixed micelles; pH sensitive. Related products Abbreviation: mPEG-NH2 Name: Methoxypoly(ethylene glycol) amine For more product information, please contact us at: US Tel: 1-844-782-5734 US Tel: 1-844-QUAL-PEG CHN Tel: 400-918-9898 Email: sales@sinopeg.com
View More