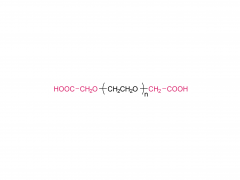
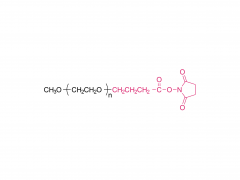
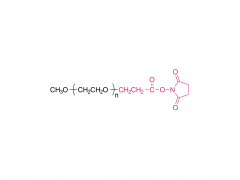
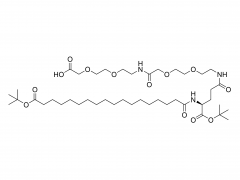
Bioact Mater. 2024 Jun 21:40:430-444. doi: 10.1016/j.bioactmat.2024.06.021. eCollection 2024 Oct. Kneadable dough-type hydrogel transforming from dynamic to rigid network to repair irregular bone defects Abstract Irregular bone defects, characterized by unpredictable size, shape, and depth, pose a major challenge to clinical treatment. Although various bone grafts are available, none can fully meet the repair needs of the defective area. Here, this study fabricates a dough-type hydrogel (DR-Net), in which the first dynamic network is generated by coordination between thiol groups and silver ions, thereby possessing kneadability to adapt to various irregular bone defects. The second rigid covalent network is formed through photocrosslinking, maintaining the osteogenic space under external forces and achieving a better match with the bone regeneration process. In vitro, an irregular alveolar bone defect is established in the fresh porcine mandible, and the dough-type hydrogel exhibits outstanding shape adaptability, perfectly matching the morphology of the bone defect. After photocuring, the storage modulus of the hydrogel increases 8.6 times, from 3.7 kPa (before irradiation) to 32 kPa (after irradiation). Furthermore, this hydrogel enables effective loading of P24 peptide, which potently accelerates bone repair in Sprague-Dawley (SD) rats with critical calvarial defects. Overall, the dough-type hydrogel with kneadability, space-maintaining capability, and osteogenic activity exhibits exceptional potential for clinical translation in treating irregular bone defects. Keywords: Dough-type hydrogel; Dynamic network; Irregular bone defect; Kneadable; Rigid network. Product: Manufacturer Of PEG Derivative By Structure,Wholesale PEG Derivative By Structure
View More