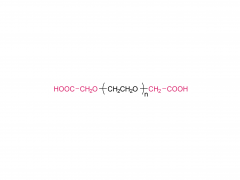
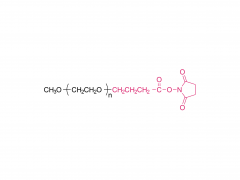
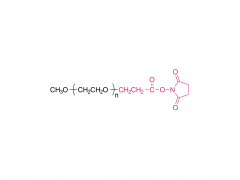
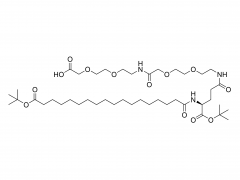
Langmuir. 2014 Jul 1;30(25):7465-74. doi: 10.1021/la500403v. Epub 2014 Jun 17. Effect of polyethylene glycol, alkyl, and oligonucleotide spacers on the binding, secondary structure, and self-assembly of fractalkine binding FKN-S2 aptamer-amphiphiles Abstract Previously we identified an aptamer, named FKN-S2, which binds the cell surface protein fractalkine with high affinity and specificity. In this paper a hydrophobic dialkyl C16 tail was added to the aptamer to create an aptamer-amphiphile. We investigated how the tail and a spacer molecule of varying length and hydrophobicity, inserted between the tail and the aptamer headgroup, affect the binding, structure, and self-assembly properties of the aptamer-amphiphile. We synthesized aptamer-amphiphiles with no spacer (NoSPR), polyethylene glycol (PEG4, PEG8, PEG24), alkyl (C12 and C24), or oligonucleotide (T10 and T5: 10 and 5 thymine, and A10: 10 adenine) spacers. The addition of the tail reduced the binding affinity of the aptamer-amphiphile over 7.5-fold compared to the free aptamer. The hydrophobic alkyl spacers resulted in the greatest loss of affinity, and the hydrophilic PEG spacers improved amphiphile affinity but did not restore it to that of the free aptamer. Interestingly, oligonucleotide spacers produced the highest affinity amphiphiles. Nucleotide composition did not affect affinity, however, as the T10 and A10 spacers had equal affinity. The oligonucleotide spacer amphiphiles had the highest affinity because the oligonucleotide spacer increased the affinity of free aptamer; the FKN-S2 aptamer plus the oligonucleotide spacer had a higher affinity than the free FKN-S2 aptamer. Circular dichroism (CD) spectroscopy and thermal melting studies indicated the aptamer forms a stem-loop and intramolecular G-quadruplex, and the tail strongly stabilized the formation of the G-quadruplex in a buffer. Cryogenic transmission electron microscopy (cryo-TEM) imaging showed the aptamer-amphiphiles, independent of the spacer used, self-assembled into micelles and nanotapes, flat bilayer structures that were often twisted. Finally, liposomes functionalized with the FKN-S2 amphiphile were incubated with fractalkine expressing cells, and the amount of binding was dependent on the concentration of the amphiphile on the liposome surface.
View More